IT Solutions
LabWare Expands Its Suite Of Available Template Solutions
Feb 15 2019
LabWare are continually expanding the number of industry-specific Solutions that are now available to LabWare customers. Industry Solutions are a unique and innovative offering that can reduce significantly the time and cost associated with implementation of a new LIMS and ELN, and that can streamline validation activities for enterprises subject to third party regulation.
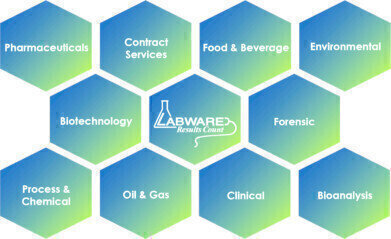
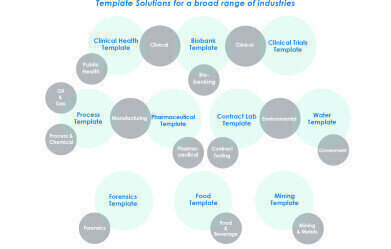
Industry Solutions are currently available for Pharmaceuticals, Contract Labs, Bioanalysis, Environmental/Water, Chemical/Petrochemical, Forensics, Biorepository, and Public Health domains.
Project metrics, gathered by LabWare over the course of numerous implementations based on use of our Industry Solutions, indicate that as much as 30% of the cost and time needed for a complete deployment may be saved through the use of this innovative and unique product offering.
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 22 2024 Marrakech, Morroco
Making Pharmaceuticals Exhibition & Conference
Apr 23 2024 Coventry, UK
Apr 23 2024 Kintex, South Korea
Apr 23 2024 Seoul, South Korea
Apr 24 2024 Jakarta, Indonesia





.jpg)









