-
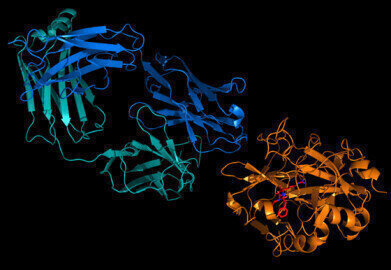 Image credit: Fab fragment of an antibody bound to thrombin. Credit: Jim Huntington
Image credit: Fab fragment of an antibody bound to thrombin. Credit: Jim Huntington
News & Views
New Spin-Out to Develop Revolutionary Drug for Thrombosis
Jun 24 2013
A new spin-out company from the University of Cambridge and Addenbrooke’s Hospital, XO1 Ltd, has raised $11 million in funding to develop a new anticoagulant drug which has the potential to save millions of lives by preventing heart attacks and strokes without causing bleeding.
The funding, from leading life science investor Index Ventures, will be used to develop ichorcumab, an antibody invented by researchers from the University and Addenbrooke’s, which targets thrombin, the enzyme responsible for blood clotting.
“This is the most exciting drug candidate I have seen in 20 years in the industry,” said Dr David Grainger, Venture Partner at Index Ventures and interim Chief Executive of XO1 Ltd. “It has the potential to save millions of lives.”
Ichorcumab is a synthetic antibody based on a naturally-occurring antibody found in a patient at Addenbrooke’s in 2008. “This patient arrived in A&E with a head injury, and we rapidly discovered a degree of anticoagulation consistent with severe haemophilia,” said Dr Trevor Baglin, Consultant Haemotologist at Addenbrooke’s, part of Cambridge University Hospitals, who treated the patient in question. “We thought it might be fatal. But to our surprise the bleeding stopped quite normally.”
The observation led Dr Baglin - and his colleague Professor Jim Huntington at the University’s Cambridge Institute of Medical Research - to design a synthetic version of the antibody in the patient’s blood that was responsible for this extraordinary anticoagulation in the hope of developing a drug which does not cause bleeding.
“Undoubtedly higher doses of these anticoagulant drugs could prevent the majority of heart attacks and strokes,” Dr Baglin explained. “But we can’t give higher doses because the bleeding they would cause would itself be fatal. Ichorcumab has the potential to change all that.”
“This antibody can deliver a high degree of anticoagulation without increased bleeding; we’ve never seen that before,” said Professor Huntington.
The investment, which comes from the $200 million Life Sciences fund Index launched last year to accelerate new drug discovery, will be used to complete the preclinical development of ichorcumab, and to manufacture substantial quantities of the antibody. “We expect to begin trials in human volunteers within two years,” said Dr Grainger.
Digital Edition
International Labmate 49.6 - Sept 2024
September 2024
Chromatography Articles - HPLC gradient validation using non-invasive flowmeters Mass Spectrometry & Spectroscopy Articles - From R&D to QC, making NMR accessible for everyone: Putting NMR...
View all digital editions
Events
Sep 22 2024 Messina, Italy
19th Confocal Raman Imaging Symposium
Sep 23 2024 Ulm, Germany
Sep 24 2024 Kielce, Poland
WoTS - World of Technology and Science
Sep 24 2024 Utrecht, Holland
Sep 25 2024 Frankfurt am Main, Germany
.jpg)

-(300-x-250-mm)_1.jpg)















