Laboratory products
Published over 3 years ago. See the latest and most current information on Laboratory products.
Every cell type in the body that makes up organs and tissues arose from a more primitive cell type called a stem cell. Stem cells are the foundation of living organisms, with the unique ability to self-renew and differentiate into specialised cell types. There are three different types of stem cell, classified by the number of specialised cell types they can produce: i) pluripotent stem cells (e.g. embryonic stem cells) can generate any specialised cell type; ii) multipotent stem cells (e.g. mesenchymal stem cells) are able to generate multiple, but not all, specialised cell types; and, iii) unipotent stem cells (e.g. epidermal stem cells that produce skin) give rise to only one cell type. It was long believed that stem cell differentiation into specialised cell types only occurs in one direction. There have been many exciting advances in stem cell biology, most notable the discovery of induced pluripotent stem cells (iPSCs) that demonstrated a mature differentiated specialised cell can be reverted to a primitive pluripotent stem cell (Takahashi K, 2006). This discovery transformed our understanding of stem cell biology enabling exciting and substantial advances in stem cell tools, technologies and applications. This article focuses on pluripotent stem cells, as they offer the most promising future applications.
To harness the power of stem cells, they must first be maintained in vitro tissue culture. Culture expansion of stem cells is tricky because they must be maintained in an undifferentiated state and not permitted to differentiate into other cell types until desired. In short, if stem cells are not dividing in log phase growth, they are differentiating. Historically, pluripotent stem cells were notoriously difficult to work with in the lab largely because of the of inherent variability of reagents derived from animal tissues.
An important concept affecting current and future innovations in stem cell technologies is Good Manufacturing Practice (‘GMP’). This is governed by formal regulations administered by drug regulatory agencies (for example the FDA) that control the manufacture processes of medicines. The use of stem cells as therapeutic agents has necessitated specialised drug regulations known as Advanced Therapeutic Medicinal Products (ATMPs). Unlike chemically synthesised medicines where the final product can be defined through chemical analysis, ATMPs are complex biological living entities whereby the entire manufacturing process defines the final product. In simple terms, every reagent that touches the stem cells in the manufacturing process throughout the entire lifetime of the stem cell becomes a component of the final product. As such, in the ‘real world’ the quality and consistency of the reagents used in a stem cell manufacturing process is paramount for downstream clinical applications, even if the project is still in the R&D or preclinical phase. Once reserved for clinical applications, GMP has become a dominating concept that affects all aspects of stem cell research and applications. Researchers and clinical developers benefit alike from GMP-focused innovations in stem cell technologies that deliver consistent growth properties and high-quality results.
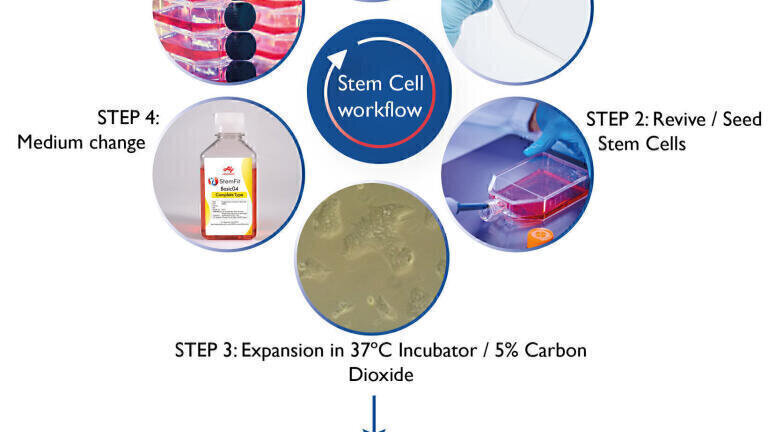
Significant advances that overcome the challenges of the past have been made in all aspects of in vitro stem cell culture. These include advances in tissue culture medium, extracellular matrix, 3D synthetic cell culture plastic, growth factors, dissociation enzymes, cryopreservation agents and differentiation technologies. The workflow to culture stem cells in vitro is not a linear process but rather a continuous circle that can be broken down into 6 steps: 1) Extracellular Matrix coating of tissue culture plasticware; 2) Revival/seeding of tissue culture flasks; 3) Expansion of the cell culture in an incubator; 4) Culture medium change; 5) Subculture or “passaging” one flask to many; and 6) Cryopreservation of the stem cell culture. The stem cell workflow is shown in Figure 1.
The art of culturing stem cells is a lot easier today than in the past. Stem cells grow as adherent cultures on the surface of tissue culture flasks or dishes (image shown in Figure 1, Step 3). For the stem cells to adhere to the surface it must be coated with extracellular matrix. In the early days, it was an effort to maintain stem cells in culture because the cultures needed to be grown on a ‘feeder layer’ of fibroblast cells. The requirement for a second cell culture combined with the stem cell culture is laborious to set up and severely limited experiments and applications (due to the contaminating fibroblasts mixed with the stem cells). Extracellular matrix isolated from mouse tumours removed the need for feeder layer cultures but can be variable in consistency and contain contaminants. Today, researchers benefit from recombinantly expressed extracellular matrix containing laminin-511 fragments that provides highly efficient adherence of a broad range of cell types and is easy to use (with only 1 hour coating time required that saves time and cost). Exceptional pluripotent stem cell adherence is achieved with laminin-511 fragments. The recombinant extracellular matrix laminin-511 is expressed in mammalian cell culture (e.g. CHO cells) or insect culture (e.g. silkworm) that eliminates the need for animal derived products in the extracellular matrix. Alternatively, synthetic 3D plastic scaffolds (e.g. Alvetex) are also available that offer a rigid defined matrix that is non-biological.
Early stem cell culture media required the medium to be replenished daily. This means 7 days a week in the lab tending to the stem cell cultures. Optimisation of tissue culture medium composition enables cultures to be maintained over the weekend without a medium change, enabling feeder-free, weekend-free stem cell culture. This may sound insignificant but does have a huge impact on the lifestyle of researchers working with stem cells. Unlike early tissue culture media, the composition of the culture media are fully defined and contain no animal derived products. Removal of animal-derived products offers important advantages by removing variability inherent in animal-derived products and guaranteeing consistent cell growth. Furthermore, animal-free formulations eleminate the risk of infection arising from the animal product (e.g. TSE risk). Growth factors are a critical component of the culture medium to maintain the stem cells in an undifferentiated state. Products available on the market contain growth factors that are expressed and isolated from barley.
Stem cells undergo cellular division in the culture vessel. As they expand, they will eventually outgrow their home and must be subcultured to separate flasks to provide space for further growth. Common practice is to use a digestive enzyme to free the stem cells from the culture surface. Trypsin isolated from bovine is commonplace in the tissue culture laboratory. Advances in the products available today use trypsin expressed in maize that is stable at room temperature in solution. Collagenase is an alternative dissociation reagent that is gentle and efficient on a wide range of cells and is available both animal-free and GMP grade - again enabling robust consistent culture conditions, and removing the dependence on animal derived products that are inherently variable.
The stem cells harvested from cultures can be frozen and stored (or ‘cryopreserved’) safely for several decades. When required, the cryopreserved stem cells may be defrosted, revived and expanded in culture providing a renewable source of stem cells. During cryopreservation of stem cells, it is critical to prevent cell death and changes in genotype/phenotype. Today’s cryopreservation media can maintain consistent high cell viability after thawing; maintaining cell pluripotency, normal karyotype and proliferation even after long term cell storage. Traditionally, the cryopreservation process involved a rate-controlled freezer or a specialised container to freeze the cells at -1ºC/min. Advances in cryopreservation agents have removed the need for rate-controlled freezing. The process is now simple - you just place the stem cell suspension into a -80ºC freezer. Moreover, cryopreservation agents are available in GMP grade and with no animal-derived ingredients.
The power of stem cells lies in their ability both to self-renew and to differentiate into specialised cell types. The process of differentiation removes the stem cells from the workflow towards applications. Directed differentiation of stem cells into specific cell types enables the number of applications to grow. A typical differentiation protocol uses stepwise changes in culture medium, cytokines, growth factors and extracellular matrix over several weeks to direct the stem cells into a particular lineage and fate. Today, innovative technologies use genetic reprogramming factors that rapidly (< 1 week) differentiate stem cells into mature cell phenotypes. This advance significantly reduces time to experiment and increases manufacturing capacity for differentiated cell types.
Table 1. Advances in Stem Cell Technologies.
Description Area of Innovation Examples of Innovative Products
Extracellular Matrix Recombinant Laminin Expressed in CHO and Silkworm iMatrix-511
Culture Medium No medium change required over the weekend, GMP grade, animal free StemFit Medium
Growth Factors Recombinant, GMP grade, animal free StemFit Purotein
Dissociation Reagents Trypsin enzyme recombinantly expressed in maize. Collagenase & Neutral Protease expressed in Clostridium histolyticum TrypLE
Collagenase NB
Neutral Protease NB
Cryopreservation Rate-controlled freezing not required. GMP grade, animal free and available for clinical use. Suitable for all cell types. STEM-CELLBANKER
Differentiation Rapid directed differentiation through genetic reprogramming Quick-Skeletal Muscle
Quick-Endothelium
Quick-Neuron
There are unlimited applications that arise from a renewable source of mature cell types. One exciting area of innovation using differentiated stem cells is in disease modelling. Studying a disease state in an organ or tissue has in the past been limited to using in vivo animal models; whereas, differentiated stem cells opened the opportunity to create disease states in specific cell types in vitro. In addition, current technologies enable organoids or ‘mini organs’ to be generated in the laboratory. Disease specific induced pluripotent stem cells can also be used to create disease models in vitro that are valuable tools for the study of disease and drug development without the need for in vivo animal models. In theory, any tissue is possible to create in vitro. In an exciting example of stem cell disease modelling, Dr Takayama from the CiRA in Kyoto, Japan has successfully modelled the life cycle of SARS-CoV-2 in both organoids and undifferentiated pluripotent stem cells (Takayama, 2020) (Sano, 2021) (Figure 2). In another example, the Skeletal Muscle Differentiation Kit was used to produce skeletal muscle myotubes from stem cells to create an in vitro disease model (Figure 3). In a direct application, pluripotent stem cell models of skeletal muscle have also been successfully used to develop a novel treatment for Duchenne muscular dystrophy (Moretti, 2020).
Promising progress is being made to create meat in the laboratory or what is commonly called ‘cultured meat’. Environmental concerns are driving the need for more sustainable meat production over traditional farming methods. Stem cell research in itself is reducing the need for the use of animals across multiple aspects as highlighted here. Producing cultured meat is straightforward in principle but faces many challenges in practice, for example maintaining the correct environment and stimuli for cultured cells to produce meat with the correct consistency and characteristics of the animal derived product. Stem cell cultures are expanded at scale in bioreactors and differentiated into skeletal muscle cells. These can be structured, using an edible scaffold for example, or used unstructured as the raw material to produce meat products (Figure 4). Tools and technologies are readily available to achieve this goal: expansion and differentiation of stem cells is highly efficient. However, a key consideration is the cost of goods. Current technologies are too costly but these are pioneering times and research is moving at an exciting pace.
The promise and potential of stem technologies to advance biology, medicine and food production can only be fulfilled if stem cell culture conditions are consistent, and accessible to research scientists and commercial operations alike. Exciting advances across multiple aspects of the stem cell workflow have streamlined processes to deliver products that are fully defined and animal-free. Furthermore, clinical translation of stem cell therapies and drug discovery are accelerated by the availability of GMP compliant reagents. The foundations are set for a bright future of discoveries and applications emerging from stem cell technologies.
Dr William Hadlington-Booth is the business unit manager for stem cell technologies and the extracellular matrix at AMSBIO. Erik Miljan, PhD, is a pioneer in the development of cellular therapies for a range of degenerative and disease conditions. He holds a PhD in biochemistry from Hong Kong University. For further information please contact:
Moretti, A. F., et al. (2020). Somatic gene editing ameliorates skeletal and cardiac muscle failure in pig and human models of Duchenne muscular dystrophy. Nature Medicine, 26, 207–214.
Takahashi K., et al. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. . Cell, 126, 663-676.
Takayama, K. (2020). In Vitro and Animal Models for SARS-CoV-2 research. Trends in Pharmacological Sciences, 41. 513-517.
Sano, E., et al. (2021). Modeling SARS-CoV-2 infection and its individual differences with ACE2-expressing human iPS cells. Iscience, 24(5), 102428.
Lab Asia 33.2 April


