Laboratory Products
PrimeStore MTM viral transport media for infectious diseases
Sep 16 2020
PrimeStore® MTM is a molecular transport media that gives the user a ‘snapshot’ in time of a sample by preserving and stabilising DNA and RNA.
PrimeStore MTM (Molecular Transport Medium) was designed and optimised for molecular testing allowing pathogenic samples to be collected, transported, and processed safely and efficiently. Millions of tubes of PrimeStore MTM have been sold to clients around the world during the SARS-CoV-2 pandemic.
The molecular transport medium inactivates infectious biological pathogens including viruses, and gram-positive/negative bacteria whilst preserving and stabilising labile DNA and RNA for downstream molecular applications.
PrimeStore MTM was invented and optimised specifically for molecular testing. In contract viral transport media (VTM) were designed for transporting samples intact to be cultured prior to the widespread use of molecular testing and contain ingredients that inhibit optimal molecular testing.
As the only FDA Class 2 cleared product for microbial nucleic acid storage and stabilisation, hospitals and laboratories trust that PrimeStore MTM provides safer and more reliable testing than UTM/VTM, saline, or research use only devices. The key differences between PrimeStore MTM and Viral / Universal Transport Mediums include:
- PrimeStore MTM does not require cold chain storage and transportation
- PrimeStore MTM inactivates the sample, along testing to take place outside of controlled containment
- PrimeStore MTM destroys enzymes and nucleases, but preserves DNA and RNA
- PrimeStore MTM does not inhibit PCR
- PrimeStore MTM has a long shelf life – 24 months, whereas as most UTM and VTM products can only claim six months
Laboratories have independently validated PrimeStore MTM with a range of platforms such as Roche, Thermo Fisher, Abbott, Siemens, Luminex, Bio-Rad, PSS, Promega, and BioMerieux, as well as the Cepheid GeneXpert point of care platform.
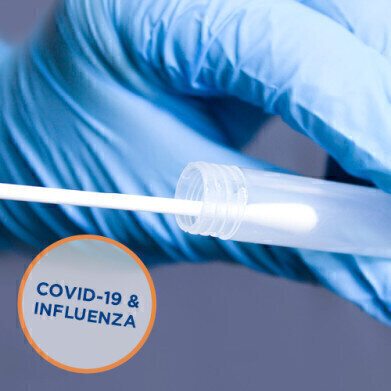
Another key feature of PrimeStore MTM is that it allows lab teams to process multiple tests from a single sample that has been inactivated and stabilised in PrimeStore MTM. This has additional advantages for testing during the upcoming flu season as both COVID-19 and flu can be tested from a single swab sample.
There are multiple peer-reviewed scientific papers, dating from 2011 onwards, supporting the use of PrimeStore MTM for pathogen detection from a wide range of clinical matrices and biofluid types collected and processed in different countries. These are available as links from the EKF Diagnostics website.
PrimeStore MTM is manufactured in the USA and UK and is available in packs of 100 tubes with a 1.5 mL fill in a choice of 3, 4 or 5 mL tubes. Tubes have external threads with leak-proof caps and can be shipped at 2-25 degrees centigrade (36-77 F).
Digital Edition
ILM 49.5 July
July 2024
Chromatography Articles - Understanding PFAS: Analysis and Implications Mass Spectrometry & Spectroscopy Articles - MS detection of Alzheimer’s blood-based biomarkers LIMS - Essent...
View all digital editions
Events
Jul 28 2024 San Diego, CA USA
Jul 30 2024 Jakarta, Indonesia
Jul 31 2024 Chengdu, China
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Aug 25 2024 Copenhagen, Denmark
-(1)-(1).jpg)


24_06.jpg)













