Laboratory Products
New Materials Testing Interface Set to Transform the Technique
Jan 22 2007
NEXYGENPlus is characterized by its extraordinary ease of use and versatility, for setting up tests, acquiring and displaying results, automating procedures, linking tests into production line control systems, exporting data to other programs and reporting results. In addition, the software includes a video capture and a unique stills capture system. Traceability and high data integrity are provided by integral security and audit trails, which allow systems to be configured to help manufacturers meet FDA 21 CFR Part 11 regulations.
With a configurable front end, NEXYGENPlus can either be used to create test protocols utilising international standards such as ASTM, DIN, EN and ISO from a library of pre-defined tests, or to generate user-specified tests.
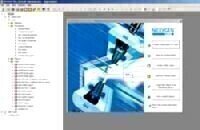
User friendliness is even further enhanced when the video or stills capture facilities are used. The video capture capability allows a camera to be linked into the system so that a close-up of the test can be displayed on the screen together with the test graph, as the test is carried out. The stills capture facility allows individual images of the sample and test jig to be captured during the test and used in post-test analysis. This also lets the operator check that the sample is mounted properly and the image can also be accompanied by some useful text information to guide the operator.
The comprehensive data handling and reporting capabilities of NEXYGENPlus allow results to be exported directly into other familiar packages such as Microsoft® Excel®, Word®, or Statistical Process Control packages. Data can be exported in a variety of formats, including CSV, TXT, ASCII and RS232. To save time, data export can also be set up to happen completely automatically - there is no need to view the results first. A variety of reporting formats can be set up for different people who may need to view the results. This lets information from a particular test or batch of tests to be reported in the most appropriate way for individuals or groups.
Digital Edition
International Labmate 49.6 - Sept 2024
September 2024
Chromatography Articles - HPLC gradient validation using non-invasive flowmeters Mass Spectrometry & Spectroscopy Articles - From R&D to QC, making NMR accessible for everyone: Putting NMR...
View all digital editions
Events
Oct 30 2024 Birmingham, UK
Oct 30 2024 Manchester, UK
Nov 11 2024 Dusseldorf, Germany
Nov 12 2024 Cologne, Germany
Nov 12 2024 Tel Aviv, Israel