Laboratory Products
New Media System for Efficient Therapeutic Protein Production
Jul 26 2023
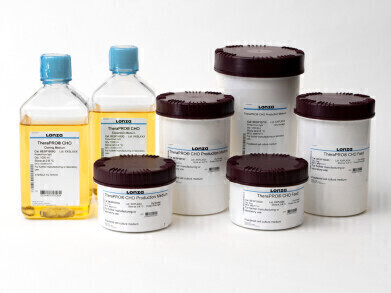
Lonza has unveiled the TheraPRO® CHO Media System, a cutting-edge cell culture platform designed to optimise productivity and improve protein quality in the production of therapeutic monoclonal antibodies. This innovative system is specifically tailored for use with GS-CHO cell lines, a prevalent mammalian host cell for protein manufacturing, offering pharmaceutical and biotechnology companies a solution to enhance product quality while expediting time-to-market.
The demand for antibody therapeutics continues to surge, driving the need for biopharmaceutical manufacturers to streamline protein production and accelerate the delivery of life-saving therapies. However, the complexities associated with protein manufacturing, as well as challenges encountered during scale-up and production, can lead to increased costs and delayed patient access.
Addressing these industry challenges, Lonza has developed the TheraPRO® CHO Media System. This advanced platform consists of a chemically defined, animal-component free, two-part production system that simplifies media preparation and workflow. Boasting full scalability, the TheraPRO® CHO Media System is supported by Lonza's team of technical experts, renowned for their extensive troubleshooting expertise. By leveraging this state-of-the-art system, biopharmaceutical manufacturers can achieve exceptional performance, including high viable cell concentrations and protein titres exceeding 5g/L during a 15-day culture cycle. These results represent more than double the protein titre achievable with existing solutions available on the market.
Every component of the TheraPRO® CHO Media System is manufactured in Lonza's ISO 13485 certified facilities, adhering to rigorous good manufacturing practices (GMP). This commitment to quality ensures full product traceability and optimum lot-to-lot consistency. Additionally, customers of the TheraPRO® CHO Media System will receive a Drug Master File (DMF) and expert technical assistance for regulatory submissions, enhancing their chances of successful approval.
More information online
Digital Edition
ILM 49.5 July
July 2024
Chromatography Articles - Understanding PFAS: Analysis and Implications Mass Spectrometry & Spectroscopy Articles - MS detection of Alzheimer’s blood-based biomarkers LIMS - Essent...
View all digital editions
Events
Jul 28 2024 San Diego, CA USA
Jul 30 2024 Jakarta, Indonesia
Jul 31 2024 Chengdu, China
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Aug 25 2024 Copenhagen, Denmark
-(1)-(1).jpg)


24_06.jpg)













