News & Views
Chemistry Testing Services Available from SSB
May 14 2017
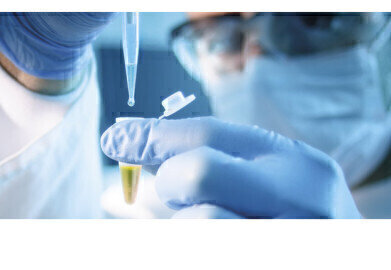
A full range of chemistry testing services available through SSB subsidiary Sartorius Stedim BioOutsource, offer characterisation of the physicochemical properties and structural attributes of therapeuticmonoclonal antibodies (mAbs).
The new service complies with the well-established ICH Q6B scientific guidelines for pharmaceuticals for human use, including methods to characterise protein structure, carbohydrate profile, post-translational modifications and impurities utilising UHPLC and LC/MS instruments. To provide these services, SSB has expanded laboratory space by 340 square meters at its facility in Glasgow, UK and has appointed new scientists with extensive chemistry testing experience. “Rapid data-driven decisions at critical stages are crucial for the successful development of biosimilars. Our chemistry testing services enable biosimilar manufacturers to benefit from side-by-side biological and physicochemical analyses to gain the in-depth characterisation data required,” explained Dr. Daniel Galbraith, Chief Scientific Officer at Sartorius Stedim BioOutsource. “Expanding our portfolio with these new services was a logical step following the successful launch of our Released N-Glycan assay in 2016,” he added.
Digital Edition
ILM 49.5 July
July 2024
Chromatography Articles - Understanding PFAS: Analysis and Implications Mass Spectrometry & Spectroscopy Articles - MS detection of Alzheimer’s blood-based biomarkers LIMS - Essent...
View all digital editions
Events
Jul 28 2024 San Diego, CA USA
Jul 30 2024 Jakarta, Indonesia
Jul 31 2024 Chengdu, China
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Aug 25 2024 Copenhagen, Denmark

.jpg)

24_06.jpg)













