IT Solutions
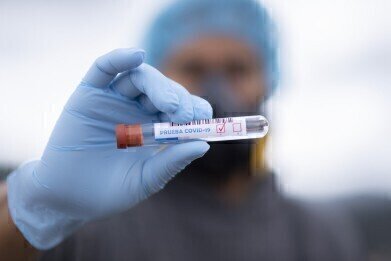
How Is COVID Diagnostic Testing Advancing?
Apr 04 2021
The outbreak of the coronavirus pandemic in December 2019 has thrown the world into disarray over the ensuing 15 months, with the scientific and medical communities scrambling to find ways to control and contain this deadly disease. A key facet of this approach has been effective diagnostic testing capabilities, which have helped to quantify the scale of the problem and allowed the authorities to take appropriate action to limit its spread.
While a rudimentary diagnostics test for COVID-19 was struck upon fairly early in the fight against the virus, the technology behind the discipline has advanced some way since then. Primarily, our capacity to refine the results that we can achieve from analysis of samples has increased, allowing us greater precision in the identification of neutralising antibodies in the immune system of the infected person. However, that’s not the only breakthrough which has been witnessed to date, with advanced automation and sample management capabilities also proving important in the struggle against COVID-19.
Precision antibody detection
From the initial (and fairly basic) diagnosis of whether a person had contracted the disease, our testing abilities have come a long way. Recently, the launch of the COVID-SeroKlir SARS-CoV-2 IgG antibody test from EKF Diagnostics has revolutionised the practice by becoming the first test capable of giving an exact analysis of the levels of antibodies in the patient. With 98.8% sensitivity and 99.6% specificity, the new test has been able to inform our understanding of the disease and how it behaves in affected persons better than ever before.
Automation
The sheer scale of patients being tested means that manually processing the number of samples has become a gargantuan task for hospitals, research centres and other medical institutions. Thankfully, the arrival of the digital transformation over the last decade has endowed scientists with the ability to automate much of the repetitive and time-consuming processes involved in diagnostic testing. This has greatly facilitated throughput and ramped up productivity, as well as providing the additional benefit of reducing human-to-human contact at a time when social distancing measures are important to contain the disease.
Sample management
As well as processing the samples involved in diagnostics, the management of them is equally cumbersome during a global pandemic. However, the advent of 2D labelling, alongside deep well microplates, has allowed researchers to get a grip on COVID-19 test samples and organise their catalogues more efficiently. This is vital at the present time, since locating, storing and sharing sample information easily and quickly is paramount to finding ways to overcome a disease that has claimed millions of lives thus far.
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
May 05 2024 Seville, Spain
InformEx Zone at CPhl North America
May 07 2024 Pennsylvania, PA, USA
May 14 2024 Oklahoma City, OK, USA
May 15 2024 Birmingham, UK
May 21 2024 Lagos, Nigeria








.jpg)






