Laboratory Products
Full 21 CFR Part 11 compliance with the most advanced rotational viscometer
Mar 11 2024
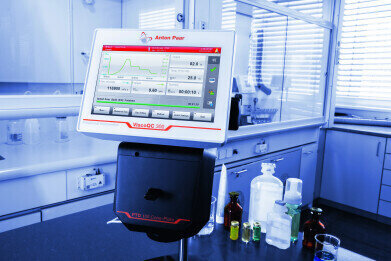
Ensuring that there is enough time is a challenge in an industry as tightly regulated as the pharmaceutical industry. Cost-effective compliance and traceability must also be strictly adhered to in order to deliver safe, high-quality products. Anton Paar’s ViscoQC 300 rotational viscometer meets and exceeds stringent standards like the FDA’s 21 CFR Part 11, and emphasises unique traceability features whilst also being able to trace and track measurements and instrument performance.
Full compliance in stand-alone mode
Activating the V-Comply software on ViscoQC 300 enables all key features to fulfill regulations of 21 CFR Part 11: audit trail (the logbook for each event); user level management and logon settings; electronic signatures with up to three-step signing process; advanced data integrity (no deletion of data, users or methods). Other useful functions include the Virtual Network Computing (VNC) mode, value visibility mode, non-storage mode and more.
The V-Comply also comes with a customised qualification package which goes far beyond simple installation and basic IQ/OQ. A detailed risk analysis, performance qualification, traceability matrix and final qualification report increase the value of the qualification package and the qualification process as a whole for the customer.
Full compliance with lab execution software
The integration with Anton Paar’s own AP Connect software, available as a 21 CFR Part 11 pharma-compliant version, further fortifies the ViscoQC Rotational Viscometer's commitment to data integrity. By facilitating paperless data management and transfer from the instrument to a central database, this connectivity minimises the risk of manual errors associated with traditional recordkeeping methods. Real-time data transfer ensures that the information is not only accurate, but also readily available, fostering a proactive approach to quality control.
Full traceability
The full strength of such applications in the regulated environment is further enhanced by the Toolmaster™ feature, i.e. automatic spindle recognition, ensuring that the instrument is not only aware of the components used, but can also restrict their use to specified methods.
More information online
Digital Edition
ILM 49.5 July
July 2024
Chromatography Articles - Understanding PFAS: Analysis and Implications Mass Spectrometry & Spectroscopy Articles - MS detection of Alzheimer’s blood-based biomarkers LIMS - Essent...
View all digital editions
Events
Jul 28 2024 San Diego, CA USA
Jul 30 2024 Jakarta, Indonesia
Jul 31 2024 Chengdu, China
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Aug 25 2024 Copenhagen, Denmark
-(1)-(1).jpg)


24_06.jpg)













