-
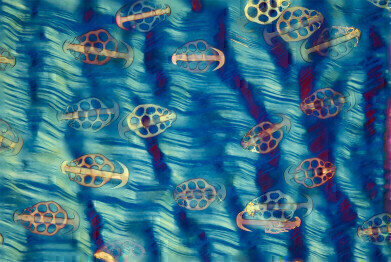 Steve Lowry - Sea Cucumber in Polarization
Steve Lowry - Sea Cucumber in Polarization -
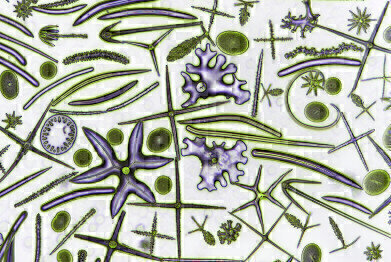 David Maitland -phase contrast image highlighting spicules of a Glass sponge
David Maitland -phase contrast image highlighting spicules of a Glass sponge -
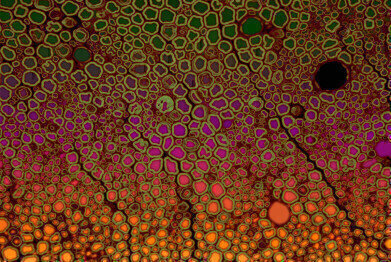 Bob Asselbergh- fluorescent image of a scietic mouse nerve
Bob Asselbergh- fluorescent image of a scietic mouse nerve
News & Views
Celebrating Art in Science
Jun 26 2018
On a quest for the best light microscopy art in 2018 and having been inspired by the beauty and breadth of images submitted for its image contest last year, admissions are now being sought for Olympus’ Image of the Year European Life Science Light Microscopy Award 2018.
Participants have the chance to win a CX23 microscope with EP50 WiFi camera and iPad, an OM-D E-M5 Mark II camera, or a PEN E-PL8 camera. The competition will run until the end of October 2018.
A jury, consisting of representatives from both science and the arts, will be judging the images. All entries will be evaluated based on artistic and visual aspects, scientific impact, and microscope proficiency.
“In microscopy you often take an image for what it shows – and then realize that it has artistic value as well,” says Dr. Christine Elstner of Olympus Europa. “What we want to achieve with the Image of the Year award is to encourage people to look at scientific images differently, appreciate their beauty and share them with others.”
Participants may upload up to three microscopy images by using the online form. Images, accompanied by a brief explanation and the equipment used, can be uploaded until 31 October 2018. The jury will select and inform the winners in November 2018.
Further details, including background information on the jury members and the full terms and conditions, can be found at www.olympus.eu/imageoftheyear
Dr Steve Lowry won the competition last year with his image showing the skin of a sea-cucumber in polarization: “The sea cucumber skin has spicules – small needle-like anatomical structures – which are paired with a plate on an anchor. The anchor articulates on the plate so it can be slightly raised. The theory is that this may help to protect the sea cucumber from predation as the anchors stick in the mouth of a potential predator and make it unpalatable. The slide is an old Victorian or Edwardian slide – from when preparing this type of specimen was very popular with microscopists. They used to arrange the plates and anchors in patterns, rather than look at a whole piece of skin. The reason I chose this image is because I had several images of arranged plates and anchors, but I was interested to look at a whole piece of skin to see the plates and anchors in their natural situation. After a long search I found a slide which had an intact piece of skin. They’re quite rare – I think I’ve only found three in my lifetime and this was the best one. The image was taken using polarised light microscopy. The sea cucumber’s skin is more or less transparent without the polarised light and I further modified the colours by using a wave retarding plate. I haven’t put extra colours in though – only those created by the microscope. I tried to reproduce it as accurately as possible. As a general rule, if I can’t do it on the microscope, I tend not to do it. When it comes to using the microscope, I find that a lot of different subjects work at the same magnification. My most used objective is the 4X objective. I occasionally go up to 10X or 20X, but I find that the lower magnification is often adequate to give you the images you want.”
Speaking about his interest in microscopy, Dr Lowry added: “I was always interested in looking at the fine details of things. When I was doing my PhD in zoology at Queen’s I was researching intertidal woodlice called Ligia oceanica to try and find humidity receptors and this got me using both light microscopes and electron microscopes. Then later at Ulster University, most of my work involved scanning and transmission electron microscopy – based on the technique, rather than on a particular subject. That means I worked on mycology, diatom research and materials science – a whole range of different topics based around electron microscopy.”
The second place award went to Dr David Maitland for his phase contrast image of a Glass sponge: “I am a zoologist and I’ve had a career teaching and researching at various universities. My interest is in invertebrate zoology, particularly form and function – I’m always fascinated by the structure and function of things. About 10 years ago I decided I wanted to perfect my photographic art, so I left academia to pursue a full-time professional photography career. When I was working in science I was taking photographs in order to illustrate lecture material and research papers. When I began my professional photography career, I was looking for a new avenue, new subjects for photography and I taught myself microscopy. I was introduced to microscopy as an undergraduate of course, but this tended to be uninspiring from a creative point of view – endless lugworm cross sections stained red turned me off! However, when I started using a microscope as an imaging tool, it opened up a new world for me. The image I took for this award is a good example of where my interests now lie – looking for the miraculous sub-structures in life and explaining why they are the way they are. The image shows the hard, circular-based structures in sponges called spicules. Sponges have soft bodies and they gain mechanical structure by embedding a scaffolding of these spicules, which also have a defence function as quite a few animals eat sponges. These are mainly spicules from the Glass sponge but there are also some from other species.
I like this image as it shows great diversity, everything you see in the picture is a spicule – they come in a variety of shapes, which tend to be characteristic of the species of sponge. As you can see from this image, the diversity is spectacular. The glass spicules come in all shapes and sizes and give sponges shape and protection against predators. Phase contrast imaging using a home-made blue phase ring highlights the three-dimensional structure of the spicules and a simple colour inversion provides the striking contrast that further accentuates these weird and wonderful shapes. Spicules are made of a form of silica, known as hydrated silica, which is in turn a form of glass chemically close to the precious stone opal.” The sample was from a fossil deposit in St Peter’s, Hungary and the slide is a Victorian museum slide.
Winning third place, Dr Bob Asselbergh used overlaying on a fluorescence image of a sciatic mouse nerve. “Even if you have beautiful microscopy pictures,” Bob says, “what ends up in a paper is usually just a very small image – or the picture doesn’t get included at all.” In his role as a research associate at the VIB at the University of Antwerp, Bob has used his expertise in microscopy and imaging to contribute to a large number of different research projects. The image Bob took his award-winning image as part of a study into nerve degeneration. “The image comes from a mouse model for peripheral neuropathy,” Bob says. “The mouse has a mutation, which is also present in humans where it causes Charcot–Marie–Tooth disease (CMT). In this mouse model we used histology to look at the sciatic nerves of the mouse. In the image you can see a cross section of these nerves stained with toluidine blue. It’s a normal light microscopy stain, from which I could measure the diameter of individual axons.”
What gave this image its striking colours was the automated process of segmentation. Using an ImageJ script designed to recognise individual axons, Bob was able to measure hundreds of axon diameters per image in a short space of time. With these measurements, Bob and his team have been able to generate powerful data to demonstrate small changes in average axon diameter. As part of the segmentation, each axon gets a unique value. Bob explains: “By applying a different colour to each value you get a new image with this beautiful gradient effect. The final image is just an overlay – a merge of the colours and the original image.” One of the effects of nerve degeneration is that the average axon diameter drops. Automated image analysis techniques measure the number and size of individual axons on microscopy images. The software also gives the image its colour gradient. The VIB-UAntwerp Center for Molecular Neurology offers a range of microscopy support to different research groups. Bob comments: “In-house we have fluorescence, live-cell imaging, laser scanning confocal microscopes as well as more simple research microscopes. We also use high-content imaging, spinning disk, super resolution and electron microscopes from the microscopy facilities of the University of Antwerp and from other VIB centres across the country.
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 28 2024 Montreal, Quebec, Canada
May 05 2024 Seville, Spain
InformEx Zone at CPhl North America
May 07 2024 Pennsylvania, PA, USA
May 14 2024 Oklahoma City, OK, USA
May 15 2024 Birmingham, UK

















