Mass spectrometry & spectroscopy
Published over 9 years ago. See the latest and most current information on Mass spectrometry & spectroscopy.
Temperate zone forests are heavily affected by climate change. Certain species like beech (Fagus sylvatica L.), common across Europe with high economic and ecological value are especially vulnerable and threatened by a retreat or possible extinction. A future task for forestry is to find varieties which can adapt to changing climate due to their efficient chemical defence mechanisms. Antioxidant leaf polyphenols represent one of these defence mechanisms. This work studied the relative quantitative evaluation of 44 compounds by HPLC-MS/MS via MRM peak areas as well as the determination of the antioxidant capacities from leaves of different beech varieties. Correlation analysis between parameters revealed that some of the most efficient antioxidant polyphenols ((+)-Catechin, Procyanidin C trimer 3, Procyanidin B dimer 4) were found in the highest levels in the leaves of the most vulnerable varieties, with the poorest growing results and also with the highest antioxidant capacities. Results showed the novel potentials of HPLC-MS/MS in the research of biomarkers of climatic adaptation in forest trees.
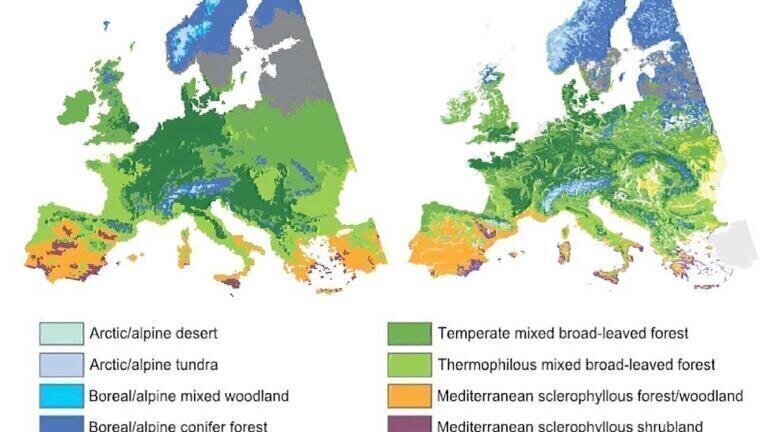
One of the apparent results of climate change is the retreat of native forests all over the world which can also result the extinction of tree species vulnerable to changed climatic conditions [1,2]. The effects of these changes on forest ecosystems, the economy as well as society are difficult to predict. European beech (Fagus sylvatica L.), which is one of the most common broad-leaved tree species in Europe and has a significant ecological and economic importance. It is especially vulnerable to increasing temperature and lower precipitation. According to recent estimations the area covered by beech forests will decrease dramatically in the next decades due to the change of climatic conditions [1,3]. Hungarian beech stands will be affected especially by these changes [4-7] which will cause beech to retreat to cooler and more humid habitats which will have unpredictable effects on forest ecosystems (Figure 1).
Figure 1. Modelled present-day (averaged for 1961–90) and future (averaged for 2071–2100) potential natural vegetation (PNV) in Europe [1].
The International Union of Forest Research Organizations (IUFRO) started an experiment in the 1990s in order to forecast future changes of beech forests in Europe: experimental forests were planted at several locations to study the growth behaviour and adaptability of different beech varieties (originating from different parts of Europe) at a given location under the given climatic conditions. In other words, to find out which are the varieties that originate from a different geographic location, but are able to adapt to the climate at the investigated location. These varieties could then provide propagating material for future afforestation’s at these locations. In the framework of the IUFRO project, an experimental forest was also planted in Hungary at Bucsuta (Zala county) in 1998. Small stands of 36 beech varieties, originating from different parts of Europe have been raised there for over 19 years (Figure 2).
Figure 2. Origin of the beech varieties grown at Bucsuta (H) in the framework of the IUFRO project.
The main question of the present article is, if there are such chemical compounds which are in a direct quantitative relationship with the degree of climatic adaptation of beech varieties, by showing a significant correlation with growth parameters (e.g. average trunk diameter). By identification and use of such chemical indicators those varieties and individuals of beech could be selected that will provide propagating material for the future afforestation and promote the preservation of beech forests in their native habitat.
One of the major defence pathways of plants is the antioxidant system. Antioxidants can be either enzymatic or non-enzymatic. Polyphenols represent one of the most important types of non-enzymatic antioxidants. The most comprehensive investigation on beech leaf polyphenols was done by Cadahia et al. [8] who have identified and characterised
43 compounds structurally by the HPLC-MS/MS technique, concluding that these compounds could be used for future studies on physiological and molecular mechanisms involved in biotic or abiotic stress in beech trees.
The present research reports on the quantitative analysis of beech leaf polyphenols in 6 selected beech varieties, grown at Bucsuta (H) using HPLC-MS/MS technique and Multiple Reaction Monitoring (MRM). Correlation analyses were conducted to reveal relationships between concentrations of individual compounds and ABTS (2,2’-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) antioxidant capacity, growth parameters as well as Ellenberg’s climatic index. By the evaluation and interpretation of relationships the most efficient antioxidant compounds were identified which could be the markers of the degree of climatic adaptation of beech. Results could contribute to selecting ‘resistant’ beech varieties that are suitable for future afforestation’s in Hungary in order to maintain native beech vegetation.
Samples: 8 trees were sampled from selected varieties (Nr. 21, 23, 26, 52, 59 and H1) during the June of 2013. 30 leaves were taken from each tree and were stored in dry ice until the extraction process.
Chemicals: Water for the extraction and chromatographic separation was produced with double distillation using conventional distillation equipment. Acetonitrile (LC-MS grade) was obtained from VWR-International (Budapest, Hungary). 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (trolox), 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid), potassium persulfate, potassium hydrogen phosphate, potassium dihydrogen phosphate and formic acid (98%) were obtained from Sigma–Aldrich (Budapest, Hungary).
Sample preparation: Leaves were treated to inactivate their polyphenol-oxidising enzymes for 2 min. with 750 W microwave energy in a household microwave oven. Leaves were ground and extracted (0.15 g leaf material with 15 ml 4:1 MeOH:H2O by stirring for 24 h in the dark). Extracts were filtered through a 0.45 µm cellulose acetate filter, and two fold dilutions of the extracts (using clean extraction solvent) were taken to HPLC-MS/MS analysis.
Chromatography conditions: HPLC measurements were carried out using a Shimadzu LC-20 type liquid chromatograph coupled with a Shimadzu SPD-M20A type diode array detector (PDA) (Shimadzu Corporation, Kyoto, Japan) and an AB Sciex 3200 QTrap triple quadrupole/linear ion trap LC/MS/MS detector (AB Sciex, Framingham, USA). A Phenomenex Kinetex C18, 150 mm × 4.6 mm, 2.6 µm core–shell column was used for the separation at 40ºC. The mobile phase consisted of A (H2O + 0.1% HCOOH) and B (CH3CN + 0.1% HCOOH). A gradient elution was run with a 1.2 mL/min flow-rate using the following time gradient: 10% B (0–1 min), 12% B (8 min), 18% B (10 min), 22% B (13 min), 28% B (19 min), 98% B (23 min), 98% B (23–32 min), 10% B (33 min), 10% B (33–40 min). 4 µl of the diluted extracts were injected. PDA detection was carried out in the wavelength range between 250–380 nm. Because of the relatively high flow rate of the mobile phase, flow-splitting was applied using a split valve, which allowed 0.6 mL/min flow to enter the MS ion source. Negative electrospray ionisation mode was used for the MS detector with the following ion source settings: ion spray voltage: −4500V, curtain gas (N2) pressure: 30 psi, spray gas (N2) pressure: 40 psi, drying gas (N2) pressure: 30 psi, ion source temperature: 500ºC. Respective MRM transitions and other optimised compound-dependent settings of the MS were used for the relative quantification of the compounds. Relative quantification involved the determination of peak areas respecting the limit of quantification (LOQ) for each compound. Measurements and evaluations were run in triplicate from each sample. Chromatographic data were acquired and evaluated using the Analyst 1.6.1 software.
Antioxidant capacity: the ABTS assay was run as described by Stratil et al. [9] at 734 nm, using the ABTS•+ radical ion and trolox standard for 10 min of reaction time. ABTS antioxidant power was given in mg equivalents of trolox/g dry leaf units (mg TE/g dw.). Measurements and evaluations were run in triplicate from each sample.
Correlation analysis: investigations and evaluation was done using the Statistica 12 software (StatSoft Inc, Tulsa, USA)
The typical chromatograms of the separation of a beech leaf extract are depicted in Figure 3.
Figure 3. The typical PDA (250–380 nm) and MRM chromatograms of a beech leaf extract. In the MRM chromatogram, different colours indicate individual MRM channels.
The amounts of individual compounds were assessed by their respective MRM peak areas instead of determining absolute concentrations. This type of evaluation was chosen because no standard compounds were available apart from (+)-catechin and (−)-epicatechin, and because some of the identified compounds showed coelution and inadequate peak resolution in the PDA chromatogram so quantification was not feasible. The MRM measurement mode of a triple quadrupole mass spectrometer provides a selective and reproducible method for quantification even if chromatographic separation could not be achieved with adequate peak resolution. By the evaluation of MRM peak areas, the composition of the extracts was compared without the need to know absolute concentrations. The characteristic MRM transitions were determined by the infusion of the extracts into the MS detector and performing MRM optimisation for each compound at its characteristic [M-H]- m/z value (Q1). Precise structural data of the 44 quantified compounds had been determined in earlier studies of the authors. The lowest limits of quantification (LOQ) were evaluated using the formula presented in Figure 4. Only those peaks were considered for the quantitative evaluation which had a peak area larger than the corresponding LOQ value.
Figure 4. Calculation of the signal-to-noise (S/N) ratio and lowest limit of quantification (LOQ) for a given MRM channel.
The peak areas indicated in Table 1 are average values of 8 individual trees of one variety. Table 1 also includes average antioxidant capacity values (ABTS) of the leaf extracts and average trunk diameters of the sample trees according to their variety. The EQ value represents the Ellenberg’s Climate quotient, which indicates if a variety originated from a site where it had adapted to dry/humid and hot/cold climate by calculating the EQ value by a simple equation in which the mean temperature of the warmest month (in ºC) is divided by the annual precipitation (in mm) and multiplied by 1000 [10].
According to Table 1 there are apparent differences between varieties respecting trunk diameter, polyphenolic composition and antioxidant capacities. As a general tendency it was observed that varieties with the poorest growth parameters (Gråsten, Torup) had the highest ABTS levels and in these varieties the concentrations of some of the compounds were also the highest (Caffeic acid-O-hexoside, Unknown 2; Quercetin-O-hexoside 1 and 2; Quercetin-O-pentoside; Kaempferol-O-pentoside) or surprisingly the lowest (Unknown 1, 3 and 6; Procyanidin B dimer 5 and 6; Procyanidin C trimer 6). From these results the following questions arise: which compounds are the most powerful antioxidants? Which compounds can take part most efficiently as antioxidants in the defence reactions of the leaves? Can certain compounds act as markers of climatic adaptation and vitality, and is there a direct and statistically provable relationship between polyphenol levels and trunk diameter? To answer these questions, a comprehensive and systematic correlation analysis was performed to reveal all relationships, including the average values of the different varieties, between concentrations of individual compounds and ABTS levels, average trunk diameters as well as EQ values.
The results of the correlation analysis are summarised in Table 2. In the case of n=6 data and p<0.05 level, the limit of significance of a correlation was |R| ≥ 0.812. It was presupposed that a significant positive correlation between ABTS antioxidant capacity levels and concentration of a given compound indicates that this compound has a strong influence on the antioxidant properties of the leaf extracts, hence it is an ‘efficient’ antioxidant compound.
According to correlation analysis, the most efficient antioxidants in beech leaf were Quercetin-O-hexoside 1 and 2, Coniferin derivative 2, (+)-Catechin,(-)-Epicatechin, Quercetin-O-pentoside, Caffeic acid-O-hexoside, Kaempferol-O-hexoside 2, Procyanidin B dimer 3 and Procyanidin C trimer 3 and 4 respecting the p<0.05 significance level. Interestingly, for some compounds significant negative correlations were indicated (R < -0.812), which could possibly be explained that these compounds have prooxidant effects in beech leaf extracts assessed by the ABTS method. Additionally, the antioxidant behaviour of isomers (especially those of Procyanidin B and C isomers) seems to be markedly different, which may be attributed to structural differences of these isomers.
By comparing the compounds’ concentrations to trunk diameter and to the EQ parameter it was shown that the varieties with higher EQ (originating from warmer and more arid regions of Europe) had lower levels of some of the most efficient antioxidant compounds by showing a significant negative correlation ((+)-catechin, Procyanidin C trimer 3 and Procyanidin B dimer 4) at the p<0.05 level. These results indicate that the varieties which were originally adapted to dryer and warmer climate do not tend to produce efficient antioxidant polyphenols in excess, as they are not ‘stressed’ in the Bucsuta region, hence their adaptability is good. These varieties also showed better growth parameters (trunk diameters) compared to low EQ varieties (see Table 1). Interestingly, some of the compounds (Procyanidin C trimer 2 and 8, Unknown 6) showed elevated levels in these varieties (significant positive correlations with the EQ value) which requires further explanation.
Respecting average stem diameter, as a direct measure of the growth and performance of the varieties it was indicated that it correlates positively with the levels of the compounds Unknown 1, 3 and 4. According to the results, these compounds can be regarded as direct leaf biomarkers of the growth and performance of beech varieties and climatic adaptation and can be used for future research of selecting varieties for beech afforestation in the investigated region of Europe. Further research will be done on the structural elucidation of the compounds labelled as ‘unknown’, which only have recorded MSn spectra, but their structure has not yet been identified.
The present study reported on the application possibilities of the HPLC-MS/MS technique for the research on polyphenolic compounds of beech leaf as potential biomarkers of the growth and climatic adaptation of beech varieties. Results indicated that there are direct relationships between concentrations of individual compounds and growth parameters of the different varieties. Although results look promising, they need to be justified by involving other varieties and investigating more sample trees per variety. Future results of the present research could be used for the selection of beech varieties for the future afforestations in Europe.
Research was supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences and by the VKSZ_12-1-2013-0034 Agrárklíma.2 project.
1. Hickler, T., Vohland, K., Feehan, J., Miller, P.A., Smith, B., Costa, L., Giesecke, T., Fronzek, S., Carter, T.R., Cramer, W., Kühn, I., Sykes, M.T. Global Ecol. Biogeogr. 2012, 21 (1), 50–63.
2. Czúcz, B., Gálhidy, L., Mátyás, Cs. Ann. For. Sci. 2011, 68 (1), 99–108.
3. Stojanoviç, D.B., Kržiç, A., Matoviç, B., Orloviç, S., Duputie, A., Djurdjeviç, V., Galiç, Z., Stojniç, S. Agric. For. Meteorol. 2013, 176, 94– 103.
4. Berki, I., Rasztovits, E., Móricz, N., Mátyás, Cs. Cereal Res. Commun. 2009, 37, 613–616.
5. Lakatos, F., Molnár, M. Acta Silv. Lign. Hung. 2009, 5, 75–82.
6. Mátyás, Cs., Berki, I., Czúcz, B., Gálos, B., Móricz, N., Rasztovits, E. Acta Silv. Lign. Hung. 2010, 6, 91–110.
7. Gálos, B., Jacob, D., Mátyás, Cs. Acta Silv. Lign. Hung. 2011, 7, 49–62.
8. Cadahía, E., Fernández de Simón, B., Aranda, I., Sanz, M., Sánchez-Gómez, D., Pinto, E. Phytochem. Anal. 2015, 26, 171–182.
9. Stratil, P., Klejdus, B., Kubáç, V. Talanta 2007, 71, 17411751.
10. Ellenberg, H. Vegetation Ecology of Central Europe, fourth ed. Cambridge University Press, Cambridge, 1988.
ILM Guide 2026/27


