Mass Spectrometry & Spectroscopy
How to Overcome LC-MS/MS Matrix Effects to Achieve Maximum Reliability
Aug 22 2018
Common approaches to counter matrix effects include matrix-matched calibration, standard addition to each sample and the application of internal standards. The first two strategies entail additional work in terms of sample preparation or additional runs, incurring further costs. Importantly, matrix-matched calibration and standard addition cannot fully compensate for matrix effects. The effectiveness of internal standards in compensating for matrix effects depends on the choice of calibrants.
Internal standards can be chemically related compounds such as derivatives (e.g. zearalanone for zearalenone) or similar compounds with identical behaviour during the ionisation process that differ only in terms of the mass of the atoms (stable isotopes). LC-MS/MS analyses rely on stable isotope dilution assays to overcome matrix effects by the addition of known amounts of stable isotope-labelled standards to the analysed sample. Stable isotope labelling involves the use of non-radioactive isotopes like ²H, 13C or 15N to replace the naturally occurring atom. Using Deuterium (²H) to replace the naturally occurring 1H doubles the mass, which is the reason why deuterated labelled standards might show retention time shifts resulting in less accurate LC-MS/MS results.
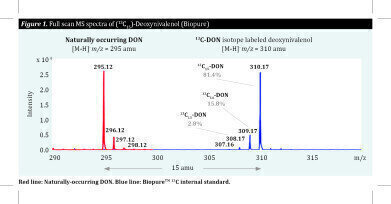
Carbon, on the other hand, is naturally present in most compounds and mycotoxins. Replacing naturally-occurring 12C by 13C changes the total mass of the atom only slightly, unlike deuterated labelled standards. Added 13C-labelled standards, e.g. 13C-labelled mycotoxins, retain the same characteristics as their 12C analogues, eluting at the same retention time from the separation column.
The difference in mass between 12C and 13C mycotoxins, as shown for deoxynivalenol in the image, allows the separation and identification of both eluted forms when the detection is performed with mass spectrometry.
The 13C peak represents the known quantity of the added standard. This peak can then be used to calculate the unknown amount of the analyte and thereby compensate for different ionisation efficiencies. In this way, innovative 13C standards can eliminate matrix effects.
Visit the Romer Labs website to learn more about 13C-labelled standards.
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 25 2024 Istanbul, Turkey
Apr 28 2024 Montreal, Quebec, Canada
May 05 2024 Seville, Spain
InformEx Zone at CPhl North America
May 07 2024 Pennsylvania, PA, USA
May 14 2024 Oklahoma City, OK, USA