Mass Spectrometry & Spectroscopy
New Range of Conductivity Reference Solutions Introduced
Nov 27 2017
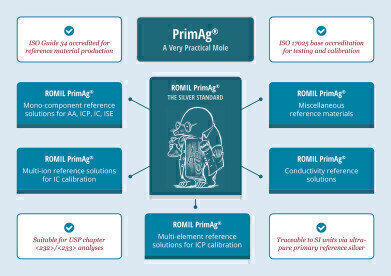
Romil is pleased to introduce PrimAg® conductivity reference solutions for the calibration of conductivity meters, further extending the comprehensive PrimAg® range of certified reference materials (CRMs). Each calibrant or reference solution is supplied with a certificate of calibration detailing its calibrated or measured conductivity, as well as the associated uncertainty traced to Romil primary silver.
PrimAg® is the clear choice when traceability is crucial. Romil is the only UK CRM manufacturer accredited to ISO 17025 requirements for calibration, and each PrimAg® product is manufactured according to ISO Guide 34 and fully traceable to SI units, eliminating the need for any additional validation. The launch of the PrimAg® range of conductivity reference solutions further extends this extensive product portfolio, perfectly complementing the existing broad selection of single and multi-element reference solutions, multi-ion solutions, pure stoichiometric substances and volumetric reagents, plus bespoke calibration mixes tailored to specific user requirements and instrumentation.
For laboratories needing to meet the forthcoming USP <232>/<233> regulations for the determination of elemental impurities in pharmaceutical products and dietary supplements, PrimAg® calibration mixes containing between six and 15 elements suitable for the calibration of inductively-coupled plasma methodologies are also available. Alternatively, laboratories can choose individually formulated reference solutions, custom-manufactured from high purity constituents characterised via the PrimAg® traceability scheme to match specific analytes or required concentrations. For maximum flexibility, there is a choice of PrimAg®-xtra, PrimAg®-plus or PrimAg®-lite – depending on the CRM – offering a range of certification levels to suit each laboratory’s specific applications.
Discover PrimAg® for yourself at www.romil.com/products/pag.
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 24 2024 Jakarta, Indonesia
Apr 25 2024 Istanbul, Turkey
Apr 28 2024 Montreal, Quebec, Canada
May 05 2024 Seville, Spain
InformEx Zone at CPhl North America
May 07 2024 Pennsylvania, PA, USA