-
 PhD student Harry Austin
PhD student Harry Austin -
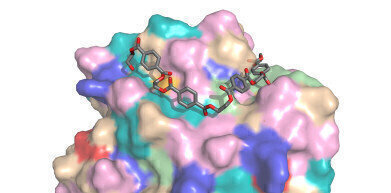 PETase - Credit H.Lee Woodcock, University of South Florida
PETase - Credit H.Lee Woodcock, University of South Florida -
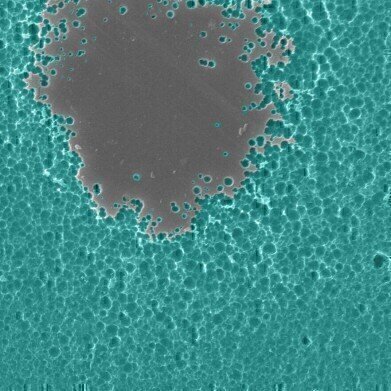 EM photos of enzyme/substate interactions. pic credit Dennis Schroeder/NREL
EM photos of enzyme/substate interactions. pic credit Dennis Schroeder/NREL -
.jpg) pic credit: Dennis Schroeder/NREL
pic credit: Dennis Schroeder/NREL -
 NREL's Donahue, Rorrer and Beckham in the lab pic credit: Dennis Scroeder
NREL's Donahue, Rorrer and Beckham in the lab pic credit: Dennis Scroeder
News & Views
Enzyme Engineering Breakthrough to Tackle Plastics Degradation
May 02 2018
In seeking a solution to the problem posed by commonly polluting plastics, research led by scientists at the University of Portsmouth and the US Department of Energy’s National Renewable Energy Laboratory (NREL), has resulted in the production of a refined version of a natural enzyme that was found to be capable of digesting plastic bottles made of polyethylene terephthalate, or PET, which currently persists for hundreds of years in the environment.
Professor John McGeehan at the University of Portsmouth and Dr Gregg Beckham at NREL made the breakthrough when they were examining the crystal structure of a natural enzyme PETase, thought to have evolved in a waste recycling centre in Japan. With help from computational modelling scientists at the University of South Florida and the University of Campinas in Brazil, the scientists used the structural 3D information to investigate features of this relatively new enzyme that suggested it might be able to accommodate man-made rather than natural polymers. This prompted the scientists to develop a mutant PETase for further study – which inadvertently turned out to be far better at degrading the plastic than the naturally occurring enzyme. They are now working on the development of the created mutant form for future industrial use.
Professor McGeehan, Director of the Institute of Biological and Biomedical Sciences in the School of Biological Sciences at Portsmouth, said: “Few could have predicted that since plastics became popular in the 1960s huge plastic waste patches would be found floating in oceans, or washed up on once pristine beaches all over the world.
“We can all play a significant part in dealing with the plastic problem, but the scientific community who ultimately created these ‘wonder-materials’, must now use all the technology at their disposal to develop real solutions. Although the improvement (to the enzyme) is modest, this unanticipated discovery suggests that there is room to further improve these enzymes, moving us closer to a recycling solution for the ever-growing mountain of discarded plastics.”
The researchers used Beamline 123 at the UK’s Diamond Light Source, one of the most advanced X-ray beamlines in the world, to generate an ultra-high-resolution 3D model of the PETase enzyme. “Being able to see the inner workings of this biological catalyst provided us with the blueprints to engineer a faster and more efficient enzyme,” Professor McGeehan said.
Diamond Light Source Chief Executive Professor Andrew Harrison, said: “With input from five institutions in three different countries, this research is a fine example of how international collaboration can help make significant scientific breakthroughs.”
Importantly, the enzyme can also degrade polyethylene furandicarboxylate, or PEF, a bio-based substitute for PET plastics that is being hailed as a replacement for glass beer bottles.
Professor McGeehan said: “The engineering process is much the same as for enzymes currently being used in bio-washing detergents and in the manufacture of biofuels – the technology exists and it’s well within the possibility that in the coming years we will see an industrially viable process to turn PET and potentially other substrates like PEF, PLA, and PBS, back into their original building blocks so that they can be sustainably recycled.”
The paper’s lead author Harry Austin, a postgraduate student jointly funded by the University of Portsmouth and NREL, said: “This research is just the beginning and there is much more to be done in this area. I am delighted to be part of an international team that is tackling one of the biggest problems facing our planet.”
The research was funded by the University of Portsmouth, NREL and the Biotechnology and Biological Sciences Research Council (BBSRC).
*Published in Proceedings of the National Academy of Sciences (PNAS)
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 25 2024 Istanbul, Turkey
Apr 28 2024 Montreal, Quebec, Canada
May 05 2024 Seville, Spain
InformEx Zone at CPhl North America
May 07 2024 Pennsylvania, PA, USA
May 14 2024 Oklahoma City, OK, USA

















