-
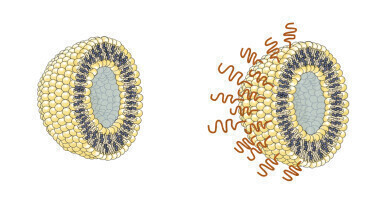 A liposome (left) containing a vaccine is commonly coated with polyethylene glycol (PEG) but it can trigger an allergic reaction to some recipients. A newly developed lipid (right) could serve as a safer, alternative liposome-coating while retaining the vaccine longer in the body. Credit: Science and Technology of Advanced Materials (2022)
A liposome (left) containing a vaccine is commonly coated with polyethylene glycol (PEG) but it can trigger an allergic reaction to some recipients. A newly developed lipid (right) could serve as a safer, alternative liposome-coating while retaining the vaccine longer in the body. Credit: Science and Technology of Advanced Materials (2022)
News & Views
Polymer Protection for Safer Vaccine and Drug Delivery
Jan 03 2023
New coating to carry vaccines and drugs into and around the body more safely
Researchers at the National Institute of Advanced Industrial Science and Technology (AIST) in Japan have developed a biocompatible polymer which could help deliver vaccines and drugs, with reduced risk of the rare dangerous adverse reaction called anaphylaxis. The preliminary tests have been reported in the journal Science and Technology of Advanced Materials.
Until now, the polymer of choice for encasing and delivering vaccines has been poly(ethylene glycol) (PEG). This synthetic, flexible, water-soluble material has been used to surround some COVID-19 vaccines carried within the tiny spherical packages known as liposomes.
Unfortunately, some recipients have suffered an anaphylactic reaction to PEG, in which the immune system mounts an allergic response to the foreign material.
The alternative polymer is a form of fatty biomolecule called a lipid, and is conjugated to 2-methacryloyloxyethyl phosphorylcholine (MPC) polymer.
This new substance spontaneously binds to the outside of liposome particles when mixed with them in water. Crucially, the polymer is not recognised by the antibodies that the body can generate in response to PEG, and tests suggest it does not stimulate any other antibodies that could cause an allergic reaction. This should allow coated liposomes containing a vaccine to be retained in the body for a longer time without being cleared by the immune system, in addition to avoiding anaphylaxis.
"We have also found that the polymer avoids other interactions with proteins in the blood that might otherwise interfere with its effects, and it also prevents liposomes from aggregating together," said Molecular Engineer Yuji Teramura of the AIST team.
Tests confirm the coated liposomes can remain stable in storage for 14 days, sufficient for real clinical applications.
"All the indications suggest that our technology should be suitable for delivering vaccines into patients who develop anaphylaxis in response to PEG," Teramura concludes.
The polymer must now be thoroughly tested in various real vaccine applications. The team is moving into this next crucial phase of the development process, prior to eventual clinical trials in humans.
More information online:
Digital Edition
Lab Asia 31.2 April 2024
April 2024
In This Edition Chromatography Articles - Approaches to troubleshooting an SPE method for the analysis of oligonucleotides (pt i) - High-precision liquid flow processes demand full fluidic c...
View all digital editions
Events
Apr 22 2024 Marrakech, Morroco
Making Pharmaceuticals Exhibition & Conference
Apr 23 2024 Coventry, UK
Apr 23 2024 Kintex, South Korea
Apr 23 2024 Seoul, South Korea
Apr 24 2024 Jakarta, Indonesia



.jpg)













